Container Closure Integrity Testing Of Lyophilized Vials – VeriPac 455
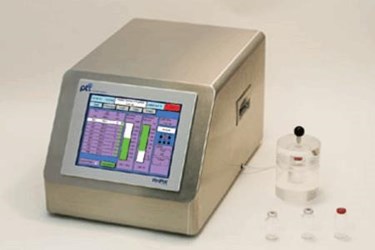
VeriPac 455 is a non-destructive inspection system used for highly sensitive micro leak detection of empty and pre-filled syringes, liquid filled and lyophilized vials and other pharmaceutical packaging formats.
VeriPac 455 is a non-destructive inspection system used for highly sensitive micro leak detection of empty & pre-filled syringes, liquid filled & lyophilized vials and other pharmaceutical packaging formats. Testing these types of products demands the highest level of sensitivity, repeatability and accuracy, and the VeriPac 455 is up to the task. It can be incorporated into protocols at any point in the handling process as it is non-destructive, non-invasive and requires no sample preparation. Applications for this technology include stability studies, clinical trial studies, quality assurance testing and production statistical process control (SPC). The VeriPac 455 is capable of detecting leak rates as low as 0.05 cc/min. Results have proven to be superior and more reliable than the dye ingress test. The core technology is based on the ASTM vacuum decay leak test method (F2338-09) recognized by the FDA as a consensus standard for package integrity testing.

