3 Emerging Trends In Drug Delivery Device Pipelines
By Selena Yu, senior oncology and hematology analyst, GlobalData

Treatment adherence is a pressing issue in patient care, stemming from lack of patient understanding of treatments, financial constraints, complexity of treatment regimens, and insufficient technical or educational support. By addressing these barriers, healthcare professionals and medical device manufacturers can ensure treatments are effective and improve patient outcomes. Since the prevalence of chronic illnesses like type 2 diabetes, chronic pulmonary disease, and cancer is rising globally, innovative methods are necessary to treat patients in a precise but also consistent manner. Concurrently, advances in microfabrication, materials science, and robotics have significantly expanded what is technically possible. The result is an emerging generation of delivery platforms that are more precise, programmable, and patient‑centric.
When analyzing the drug delivery pipeline a few themes stand out: patient accessibility, precise drug delivery, and painless administration. Current technologies that support these themes include microneedle patches, robotic pills, and biodegradable devices.
Microneedling: Patient Accessibility
Microneedle patches revolutionize transdermal drug delivery. Patches are pain-free, reduce risk of infection, and are easier for healthcare professionals or patients to administer when compared to traditional intravenous methods. Compared to traditional transdermal patches, microneedles allow drugs (including larger molecules and biologics) to penetrate beyond the skin’s outer barrier, which results in increased bioavailability. Microneedle patches often produce comparable pharmacokinetic profiles for many compounds when compared to injectables, without deep tissue penetration.
For example, Daewoong Pharmaceutical is currently in clinical trials with its closed-pack aeropressured microneedle patch (CLOPAM) for administration of GLP-1 analogues, like semaglutide.1 The company’s research has showed that the relative bioavailability is around 80% when compared to standard injection. In other words, when subcutaneous injection is the route of administration, drug absorption is considered 100%.2 Under the same conditions, microneedle patches achieved over 80% drug absorption. 2 This is the highest concentration observed compared to existing microneedle patches containing the same active ingredient, which showed a bioavailability of around 30%. To further compare, drug absorption with CLOPAM is approximately 160 times higher than that of the oral semaglutide formulation. CLOPAM, and other microneedle patches, are more effective than current oral formulations and have the potential to make semaglutide more accessible to patients by eliminating injections and thereby injection-related fear and discomfort.
Another area of therapeutic advancement of microneedle patches is in vaccine delivery. A device, the Micron Microarray Patch, is in clinical development by Micron Biomedical.3 This peel and stick patch is designed to reduce the need for a cold chain and allows a community health worker to vaccinate children within minutes. The technology is applied to the skin and, by pressing a button, there is confirmation of administration. This device will help rural communities in low- and middle-income countries gain access to lifesaving vaccines. It has been designed against viral infections that mainly impact children, like rubella and measles.
Robotic Capsules: Painless Administration
Robotic capsules are another emerging technology in drug delivery. Robotic pills are ingestible devices designed to navigate the gastrointestinal tract and mechanically deliver drugs through the intestinal wall (transenteric injection). This technology has the ability to increase bioavailability of certain drugs that lose efficacy when administered in an oral pill form. Therapeutically, the treatment is analogous to a self‑administered gastrointestinal‑based injection, while the patient experience is simply taking a standard oral capsule. If efficacy, safety, and reliability data ultimately mirror that of subcutaneous administration, such pills could significantly reduce barriers to starting and maintaining biologic therapy, improving adherence, and potentially expanding the eligible patient pool.
Rani Therapeutics is currently in clinical trials with their device, the Rani Pill, to validate efficacy of orally delivered large molecules including peptides, proteins, nucleotides, and antibodies.4 The pill navigates through the stomach and small intestine and once it has positioned itself, a small needle injects the drug into the intestinal wall. The formulation ensures that the drug stays protected within the pill until it is localized for injection.
In a recent canine trial, the Rani Pill, administering semaglutide, was well tolerated and demonstrated bioavailability and biological activity of 107%,5 when compared to subcutaneous administration. Drug administration via transenteric injection allows for localized release (pH dependent) and rapid absorption, all while remaining virtually painless to the patient.4 With the positive results from their trial, Rani Therapeutics could utilize the Rani Pill platform to partner with pharmaceutical companies to develop oral versions of its injectable drugs or enhance the bioavailability of existing oral drugs.
Biodegradable Drug Delivery: Precise Drug Delivery
Biodegradable drug delivery devices offer patients multiple benefits, from fewer complications to a more environmentally friendly profile. This method has been used for precise drug delivery in cancer and ophthalmologic diseases. For example, ChemoSeed, created by CRISM Therapeutics, is an implantable biodegradable drug delivery device designed to deliver irinotecan directly into cancer tissues for treatment of resectable glioblastoma.6
In the field of ophthalmology, a hydrogel scaffold technology manufactured by GelMEDIX is designed to deliver induced pluripotent stem cell-derived retinal pigment epithelial cells to restore vision in late-stage geographic atrophy directly to the ocular surface.7 Instead of needing to remove this implant after, the therapy-loaded hydrogel biodegrades, which not only ensures that the full dosage was received but also increases patients’ comfort while receiving treatment.7
EyePoint Pharmaceuticals’ EYP-1901 is being evaluated in two Phase 3 clinical trials (NCT06683742 and NCT06668064). It is a bioerodible implant designed to release a tyrosine kinase inhibitor (TKI), vorolanib, into the subconjunctival space of the eye for treatment of retinal diseases such as wet age-related macular degeneration, diabetic retinopathy, diabetic macular edema, and retinal vein occlusion. Regeneron’s AXPAXLI is also in clinical trials (NCT07235085, NCT06223958, NCT06495918). It is a bioresorbable hydrogel implant that uses a similar sustained-release of a TKI, axitinib, making it comparable to EYP-1901, as it is being studied for treatment of the same set of retinal diseases.
Biodegradable devices can improve patient compliance by reducing frequency of administration. By engineering the material’s degradation rate, the medication can be released gradually, over an extended period. Additionally, with the therapeutic target at specific sites of administration, drug efficacy is maximized, while limiting off-target toxicity. Because of their biodegradability, the products have increased biocompatibility and safety since they are broken down into nontoxic byproducts, eliminating the risk of long-term complications, like chronic inflammation, which are associated with traditional nondegradable devices. Further, patients avoid a removal surgery once the implant has completed drug administration.
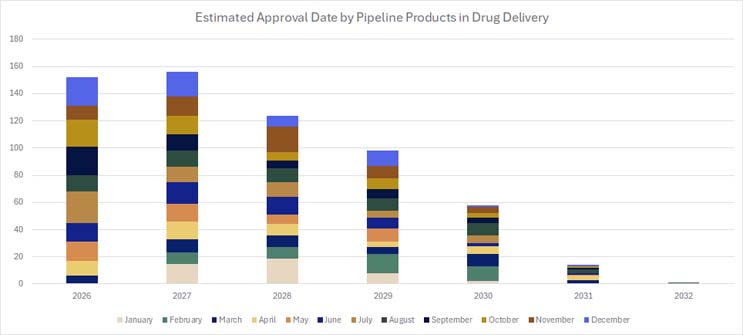
When Will These New Devices Hit The Market?
Many devices are projected to be approved in the next one to three years, meaning we may see them hit the market within three to five years (Figure 1).8 New technologies and pharmaceuticals entering the market inspire more innovation in this space.
Figure 1. Click on image to enlarge.
There is a focus on creating methods outside of injections specifically for GLP-1 agonists. Several drug delivery devices in the pipeline, like Rani Pill and CLOPAM, have gone through additional trials or design changes to accommodate recently popularized GLP-1 analogues like semaglutide. Further, new indications for the drug are actively being studied and approved. For example, Ozempic was approved for kidney disease in January 2025 and is currently in clinical trials for a peripheral arterial disease/peripheral vascular disease indication.
Broadening the scope of indications increases the number individuals who would benefit from the drug, thus increasing the population of patients who need another form of administration over the standard injection. The technology used to develop more efficient drug delivery for GLP-1 agonists is translatable to other drug modalities as well, expanding drug delivery modes across different disease areas.
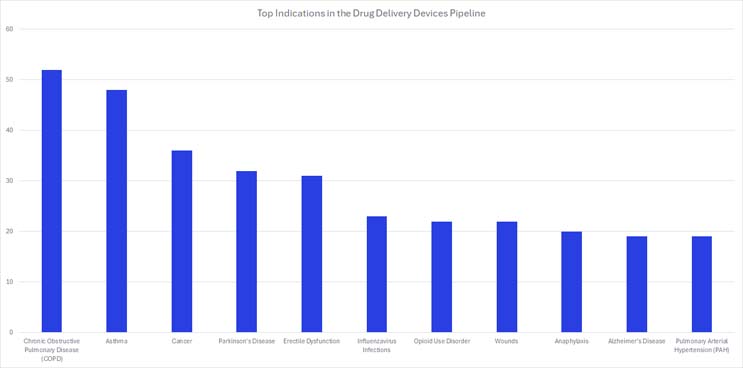
Top Indications In Drug Delivery Device Product Pipelines
When analyzing 950 unique pipeline drug delivery device products, the top indications other than COVID-19, were chronic obstructive pulmonary disease (COPD), asthma, and cancer (Figure 2).8 The COPD and asthma devices in development are primarily inhalable devices designed to deliver aerosolized medications, comparable to existing products on the market.
Figure 2. Click on image to enlarge.
On the other hand, devices used to treat cancers differ vastly from one another because of the different manifestations of the disease, leading to more innovation in the space. Many of the devices currently on the market are injectable or infusion type devices. A few interesting products in cancer care focus on localized drug delivery and easy administration like minimally invasive implantable devices and patches. The top pharmaceutical companies with devices in their pipelines are CF Pharmatech, LTT Bio-Pharma Co, Lupin Pharmaceuticals, and Sinsin Pharmaceuticals.
Beyond The Pill: Moving Forward
The drug delivery device sector has seen a marked shift in development, centered around:
- patient accessibility
- precise drug delivery
- painless administration
- reformulation of current products
- improvement of drug absorption and efficacy.
Microneedle patches, robotic pills, and biodegradable devices innovate drug delivery by increasing drug bioavailability and biological activity without requiring continual reformulation of drug products. They streamline pharmaceuticals by taking a treatment that once required a patient to sit through an injection or infusion to a product focused on patient accessibility, encouraging effective treatment management.
Not only do these devices improve the patient’s treatment journey, but they also increase likelihood of treatment adherence and, ultimately, patient outcomes. From the perspective of developers, when adherence increases, so does the ability to bolster portfolios and capitalize on partnerships that produce more sophisticated drug delivery devices and systems.
References
- Daewoong Therapeutics Inc. CLOPAM Microneedle Platform. https://en.daewoongrx.co.kr/RND_microneedle [Accessed March 10, 2025]
- PR Newswire. Daewoong Therapeutics Microneedle Patch Achieves Best-in-Class Bioavailability, Proving the Strength of Its Drug-Delivery Platform. https://www.prnewswire.com/news-releases/daewoong-therapeutics-microneedle-patch-achieves-best-in-class-bioavailability-proving-the-strength-of-its-drug-delivery-platform-302528778.html [Accessed March 10, 2025]
- Micron Biomedical. Technology. micronbiomedical.com/news-type/press-releases/ [Accessed March 10, 2025]
- Rani Therapeutics. Technology. https://www.ranitherapeutics.com/technology/ [Accessed March 10, 2025]
- Rani Therapeutics. Rani Therapeutics Announces Preclinical Data Demonstrating Successful Oral Delivery of Semaglutide via RaniPill®HC. https://ir.ranitherapeutics.com/news-releases/news-release-details/rani-therapeutics-announces-preclinical-data-demonstrating/ [Accessed March 10, 2025]
- CRISM Therapeutics. https://www.crismtherapeutics.com/chemoseed/ [Accessed March 10, 2025]
- GelMEDIX. Technology. https://www.gelmedix.com/our-technology [Accessed March 10, 2025]
- GlobalData Medical Intelligence Center, Pipeline Products Database [Accessed March 16, 2025]
About The Author:
 Selena Yu is a senior oncology and hematology analyst at GlobalData. With a background in microbiology and immunology, she began her career in market research, spending four years focused on diagnostics and drug delivery. She has recently moved to GlobalData’s pharma oncology team, where she supports analysis of oncology pipelines, markets, and competitive landscapes. Before joining GlobalData, Yu completed her MSc at McGill University in microbiology and immunology, researching therapeutic and vaccine candidates for schistosomiasis, a neglected tropical disease.
Selena Yu is a senior oncology and hematology analyst at GlobalData. With a background in microbiology and immunology, she began her career in market research, spending four years focused on diagnostics and drug delivery. She has recently moved to GlobalData’s pharma oncology team, where she supports analysis of oncology pipelines, markets, and competitive landscapes. Before joining GlobalData, Yu completed her MSc at McGill University in microbiology and immunology, researching therapeutic and vaccine candidates for schistosomiasis, a neglected tropical disease.