New Wound Closure Technology Set To Triple In Value By 2025, Cannibalize Competitors
By Mikayla Schaffner and Sean Collins, iData Research

Traditionally, surgical wounds have been closed with sutures, staples, Steri-strips, Mastisol, or some combination thereof. There are multiple benefits to using these technologies, but new products are expected to significantly improve the way surgeons close wounds. Ethicon’s Dermabond Prineo mesh and cyanoacrylate-based skin closure systems, as well as Zipline Medical’s Zip Surgical Skin Closure, are new products that recently have become popular for surgical wound closure. This rise in popularity is cannibalizing older forms of wound closure, such as sutures, staplers, and skin closure strips.
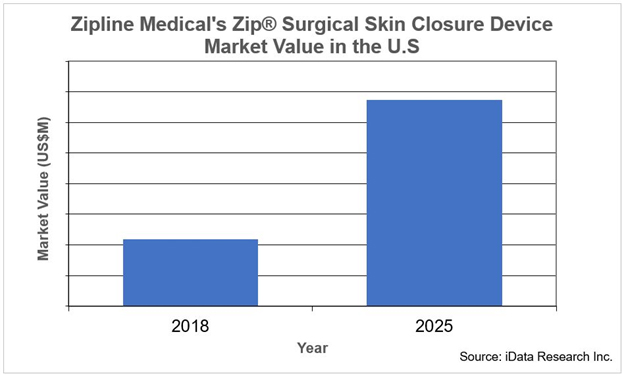
Zip Skin Closure Set To Triple In Value By 2025
Zipline Medical’s Zip Surgical Skin Closure device is primarily used to close surgical incisions. Unlike staplers and sutures, this product is less invasive and less traumatic for patients when being removed. In addition to improved comfort, the device requires less time to close the wound, and the scar is less pronounced. In the past year, the number of Zip Surgical Skin Closures used has more than doubled.
This trend will continue, with the number of Zip Surgical Skin Closures used nearly tripling by 2025. While this specific “zip style” technology is patented, similar wound closure systems using clips, loops, or adhesives have been explored; they have not achieved the same success. This market is quite new, so growth rates are high, but even as it matures and growth rates slow, this innovation proves the wound closure market holds great potential.
In addition, this technology has been implemented into consumer products for use in closing small lacerations that do not require a doctor’s visit. Currently, there is only one company participating in this emerging market — Zipline Medical, with its ZipStitch product — but as companies begin to see the value of these types of closure devices, they may begin to market similar products.
New Sizes Drive Sales Of Ethicon’s Dermabond Prineo
Ethicon’s Dermabond Prineo mesh and cyanoacrylate-based skin closure system plays a similar role to Steri-strips or Mastisol, but causes fewer reactions, is completely waterproof, and protects the wound from infection. As a newer product, Dermabond Prineo’s market value has steadily increased each year as surgeons begin to adopt this technology. This is a single-player market, so other manufacturers have plenty of opportunity to take advantage in its early stages.
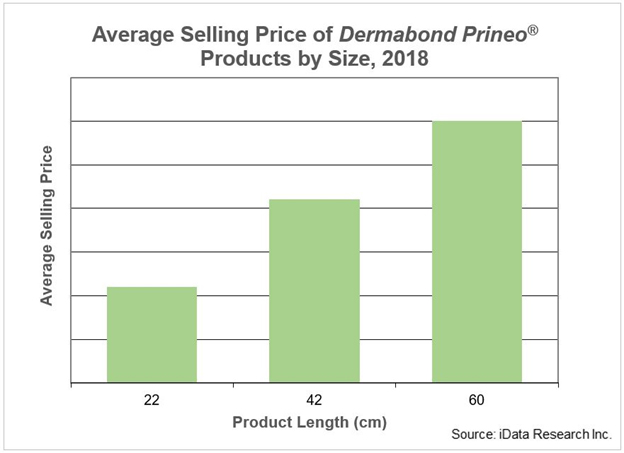
Ethicon released its first Dermabond Prineo product in 2011. It was a 60-cm product that surgeons could cut into different sizes, depending on the length of the wound. Since then, Ethicon released a 22-cm product in 2014 and a 42-cm product in 2018. In 2018, the 22-cm product was the most popular, but 42-cm product sales are increasing rapidly. Although the smaller, newer products are becoming more popular, the 60-cm product still makes up a significant portion of sales.
Trends Driving This New Technology
The wound closure market has begun to adopt these new technologies for surgical incision closure. It is expected that, in the future, zip skin closure and similar products will take market share away from the suture and stapler market. Similarly, the mesh and cyanoacrylate- based skin closure system is predicted to cannibalize the skin closure strip market. These new products have opened the door for innovation among companies as they attempt to compete for the most efficient way to close surgical incisions.
The benefit of these new closure technologies compared to previous options is immense, and once surgeons begin using them, they are likely to continue. They are easier to remove and do not puncture the skin, decreasing the risk of infection. In addition, these devices reduce the duration of surgery and the time required to heal a surgical incision. Although these products have an increased cost, they will still save hospitals money by decreasing surgery time — resulting in pressure from patients, surgeons, and hospitals to use these new devices, thus driving the market.
As more competitors enter these emerging markets, competition is expected to erode prices. Lower prices will drive the adoption of mesh and cyanoacrylate-based and zip skin closures as they become more similarly priced to older products. These new wound closure markets have the highest growth rates within the overall wound and tissue management market — driving the market overall, but doing so at the expense of the suture, skin stapler, and skin closure strip markets.
Nonetheless, growth in surgical procedure volume will cause the value of the total wound closure market to appreciate over the forecast period. In 2018, the wound closure market was valued at over $3 billion, and it is projected to approach $5 billion by 2025. Overall, the total wound and tissue management market is expected to grow at a CAGR of 4.7 percent, reaching approximately $15 billion by 2025, primarily driven by an increase in obesity rates, chronic diseases rates, and a growing elderly population in the United States.
References
- United States Wound and Tissue Management Market Report Suite 2019 – MedSuite, iData Research
- MedSKU - Zipline Medical Zip Surgical 2018, iData Research
- MedSKU - Ethicon DERMABOND PRINEO 2018, iData Research
About The Authors
Mikayla Schaffner is a Research Analyst at iData Research and was the lead analyst on the United States Wound and Tissue Management Market Report Suite 2019 – MedSuite. In addition to studying the U.S., her work expands out into Europe and Asia.
Sean Collins is an Analyst Manager at iData Research. He has served as the principal analyst for dozens of medical device research projects, syndicated and consulting, and now leads others in doing the same.
About iData Research
iData Research (http://www.idataresearch.com) is an international consulting and market research firm, dedicated to providing the best in market intelligence for the medical, dental and pharmaceutical industries. Our research empowers our clients by providing them with the necessary tools to achieve their goals and do it right the first time.
