Reverse Engineering A Human Heart Valve
By Jim Pomager, Executive Editor
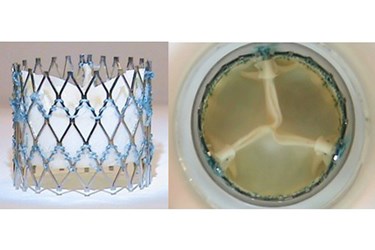
The aortic valve is a remarkable piece of biomechanical engineering. On any given day, the leaflets (or cusps) of a healthy aortic valve will open and close 100,000+ times, allowing the proper amount of blood to flow from the heart to the rest of the body. Over a lifetime, a healthy valve endures more than 3.4 billion heartbeats.
Unfortunately, the aortic valve doesn’t always remain healthy. (What organ does?) According to the American Heart Association, up to 1.5 million people in the United States suffer from aortic stenosis (AS), a calcification of the aortic valve that narrows its opening and restricts blood flow. In the early stages, the disease is often asymptomatic, but as it progresses, it can cause chest pain, weakness, and difficulty breathing. And in approximately 300,000 people worldwide, the condition develops into severe AS, which has a one-year survival rate of approximately 50 percent, if left untreated.
Fortunately, there are treatment options. The most common and successful is aortic valve replacement (AVR), wherein a mechanical or tissue-based valve is substituted for the diseased valve. For decades, replacement valves were implanted via open heart surgery, which involves an extended hospital stay and months of recovery. But in recent years, a promising new approach has emerged: transcatheter aortic valve implementation (TAVI), also known as transcatheter aortic valve replacement (TAVR). In TAVI, a tissue-based artificial valve is delivered into the diseased heart valve via a blood vessel, rather than through a large incision in the chest.
TAVI has many benefits, the most obvious (and compelling) of which is its noninvasiveness, which means shorter recovery times and faster attainment of quality-of-life outcomes for the patient. Replacement of a transcatheter aortic valve (TAV) can also be a minimally invasive exercise — a second TAV can simply be implanted within the first.
On the other hand, the use of TAVI procedures in U.S. hospitals is not yet widespread (though it is growing rapidly). The longevity of current-generation TAVs also remains unknown because it is an emerging technology, compared to evidence of 15+ years for surgically implanted heart valves. Plus, TAVI is only approved in the U.S. for use in AS patients who are either ineligible for surgical valve replacement or at high risk. (TAVI has been available in Europe since 2007, and clinical trials are underway in the U.S. for its use in intermediate-risk patients.)
What’s really needed is an improved TAV — one that outperforms current transcatheter valves, is as durable as a surgical valve, and operates more like … well, a healthy human aortic valve. Such a valve would open the door to TAVI’s use in the hundreds of thousands of lower-risk (and generally younger) AS patients whose only current option is a surgically implanted valve, and who would rather not have their chest opened.
Now, a man who has dedicated his professional career to studying the aortic valve has invented a new artificial valve design that he says will revolutionize TAVI. And if everything goes according to plan, his TAV will reach European patients in 2015 and U.S. patients soon after. How did he and his startup company design such technology? By reverse engineering the aortic valve.
The Man Behind The Valve
 Mano Thubrikar, quite literally wrote the book on heart valves and heart disease — two of them, in fact. His The Aortic Valve (1989) and Vascular Mechanics and Pathology (2007) are leading textbooks in cardiovascular studies, and the former is widely used as a guide in the design of bioprosthetic heart valves.
Mano Thubrikar, quite literally wrote the book on heart valves and heart disease — two of them, in fact. His The Aortic Valve (1989) and Vascular Mechanics and Pathology (2007) are leading textbooks in cardiovascular studies, and the former is widely used as a guide in the design of bioprosthetic heart valves.
After earning an undergraduate degree in metallurgy, a master’s in materials science, and a Ph.D. in biomedical engineering, Dr. Thubrikar spent the first 30 years of his career exclusively in academic research. He studied the aortic valve and bioprostheses from almost every conceivable angle while working at the University of Virginia (UVA) and at the Carolinas Medical Center and the University of North Carolina (UNC) at Charlotte.
But in 2003, Dr. Thubrikar received a phone call that would change the trajectory of his career and set him on the path to develop a novel TAV technology. A woman contacted him to discuss her son, a 35-year-old athlete with a calcified aortic valve. The condition was the result of a bicuspid valve, a congenital condition where the aortic valve has two cusps, rather than the customary three. The man needed a valve replacement, and his only choice was to have a mechanical heart valve surgically implanted. However, the surgical valve meant he would have to stay on anticoagulants for the rest of his life, effectively ending his athletic pursuits. Dr. Thubrikar informed the mother that there just weren’t any treatments available that would allow her son to continue his active lifestyle.
“Didn’t you write the book on the aortic valve?” she asked. “Why didn’t you make a valve that my son could use?”
The conversation and question deeply affected the researcher. “I went home and was so disturbed,” he told me during a recent visit to his office. “I talked to my wife and said, 'You know what? Years of research, writing papers, and giving presentations — that’s done. I now need to make a heart valve.'”
Soon after, Dr. Thubrikar left Carolinas Medical Center to embark on his new mission. He joined artificial heart valve pioneer Edwards Lifesciences as a Distinguished Scientist, but left after it became clear that the company's plans for him didn’t align with his own.
So in 2007 — coincidentally, the same year Edwards launched the first commercially available TAV device — Dr. Thubrikar returned to academia, joining the staff at the South Dakota School of Mines & Technology. There he spent the next three years working on a new artificial valve design — one based on decades of research on the physics behind the human aortic valve.
Looking To The Human Body For Design Output
According to Dr. Thubrikar’s research, the natural aortic valve follows four strong design principles for maximum longevity and optimal hemodynamic performance. Those criteria are:
1. A specific coaptation height — When the valve’s three leaflets come together to close the valve, there is some surface-to-surface contact between the leaflets, rather than an edge-to-edge seal. This safety margin helps prevent against blood leakage back into the left ventricle.
2. No folds in the leaflets — Natural aortic valve cusps flex without folding. Folds would crease the tissue and cause unwanted stress on the leaflets, negatively impacting durability.
3. Minimum overall height — Extra height would produce dead space, which can lead to a variety of issues.
4. Minimum leaflet flexion — The human aortic valve manages to open completely with the leaflets moving only 70 degrees, not the 90 degrees you might expect. Again, this improves the valve’s longevity.
“You almost need to be a solid geometry design engineer to understand the math and the equations behind these principles,” he explained. “With these criteria, however, you have design parameters for the aortic valve. The mathematical equations give you the output of how an artificial valve should be designed.”
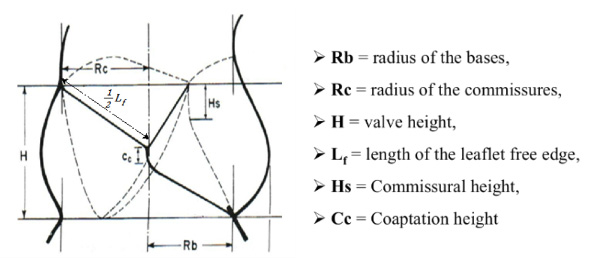
Dimensions of the natural aortic valve
Based on these four principles, Dr. Thubrikar reverse engineered the aortic heart valve, developing a new artificial valve design that mimics the aortic valve’s precise geometry. In October 2010, he launched a startup company called Thubrikar Aortic Valve, Inc. to commercialize his new creation, which he calls Optimum TAV and touts as “nature’s valve by design.”
“When someone asks me, ‘How does your valve compare with Edwards’?’ or ‘How does your valve compare with Medtronic’s?’, I say ‘We don’t compare our valve to them,’” Dr. Thubrikar told me. “We compare our valve with the natural aortic valve.”
On the surface, Optimum TAV looks similar to other artificial heart valves on the market, with three leaflets of bovine pericardium tissue mounted on a metal stent-frame. (In fact, the design is often mistaken for another widely used surgical valve.) But according to Dr. Thubrikar, it has a unique combination of features that will help it overcome the major design limitations of current-generation TAVs (if we’re going to compare). Those design limitations include:
- Suture holes in the leaflet body — While all TAVs (including Optimum TAV) are constructed by sewing animal tissue to a metal frame, piercing the flexion zone of the leaflets leads to potential wear. Optimum TAV does not have a single suture hole in the working portion of the leaflet body.
- Blood flow through frame — Some TAV frames are as tall as 5 cm in height, extending up into the aorta once implanted. As a result, blood must pass through the frame to enter the coronary arteries. Proteins in the blood will accumulate on the frame, and can eventually break loose and cause thromboembolisms (blood clots). Optimum TAV is only 2 cm in height. (Related, the low height of the Thubrikar valve also makes it less likely to require a pacemaker.)
- Thick outer frame — The thicker the frame, the smaller the valve opening will be, allowing less blood to pass through. This opening is referred to as the valve’s EOA, or effective orifice area. The average EOA of a surgical valve is around 1.9 cm2, and some TAVs have EOAs as small as 1.5 cm2 (technically, a mild form of stenosis). In bench tests, Optimum TAV’s EOA was 2.3 to 2.4 cm2. (A healthy aortic valve has an EOA of approximately 2.7 cm2.)
- Clipped calcified leaflets — Some current TAVs are anchored to the patient’s original valve using a paper-clip like mechanism. In this design, there is the potential that the TAVs leaflets will come into contact with the old, calcified leaflets during the operation, causing wear. Optimum TAV’s design eliminates the possibility of contact between the leaflets and native valve.
- Paravalvular leakage — In some cases, a space forms between the outside of a TAV and the surrounding heart tissue, and blood can leak through. Optimum TAV has a high skirt to prevent this type of gap from developing. In addition, Optimum TAV’s novel frame architecture allows it to conform to and seal off either a round or elliptical annulus (the ring-shaped base of the original valve). This is particularly helpful in minimizing or eliminating leakage in bicuspid patients, who often have an irregularly shaped annulus.
- Balloon expansion — TAV frames made of stainless steel must be forced open by a balloon. The TAV’s tissue can get caught between the balloon and the frame and potentially tear. Optimum TAV’s frame is made of nitinol, which automatically expands once deployed from the catheter.

Optimum TAV
“Other technologies have built-in issues,” Dr. Thubrikar said. “To be able to avoid those problems in a comprehensive fashion is no small feat.”
Trial By Fire
During the two and a half years following the establishment of Thubrikar Aortic Valve, Optimum TAV seemed to be moving steadily toward market. The company raised enough funding to get started, primarily from friends, family, physicians, entrepreneurs, and technology industry executives. Patent applications were filed, suppliers were selected, valves were painstakingly produced (by hand, over one-and-a-half to two days each), and preclinical testing began.

Members of the Thubrikar Aortic Valve team (left to right): Deodatt Wadke, member of the board of directors and cofounder; Samir Wadke, executive director of business development and cofounder; Dr. Mano Thubrikar, president and founder; Samuel Evans, research engineer II; and Nikhil Heble, counsel, secretary, and cofounder
But the fledgling company was dealt a major setback in April 2013, when a fire destroyed the Horsham, Pa. office building to which the Thubrikar Aortic Valve laboratory had recently relocated (from South Dakota). All of its equipment was destroyed and needed to be replaced. The company had to relocate to nearby Norristown, Pa. Not an ideal scenario for a startup trying to make the most of extremely limited resources.
The company was undeterred by the fire, and the last year has been a successful one for Thubrikar. The company completed most of its preclinical testing (including implants in 12 animals and two diseased human cadaver hearts), reached design freeze on Optimum TAV, filed a provisional patent application for its proprietary delivery catheter, and achieved almost $2 million in total funding. Perhaps the biggest milestone came in August 2013, when Optimum TAV met the International Organization for Standardization’s (ISO’s) durability requirements by surpassing 200 million cycles in a third-party ISO certified laboratory.
The durability testing has continued, and Optimum TAV continues to function beyond 390 million cycles, which approximates 11 years in vivo. Surgical valves typically last anywhere from 12 to 18 years, and Thubrikar expects his valve to last at least that long.
“I would not be surprised if it surpasses the longevity of even the surgical valve,” he said.
The company also received its first institutional investment, from Delaware Crossing Investor Group (DCIG), in 2014. The primary DCIG investor, Marv Woodall, led the commercialization of the world’s first stents as president of Johnson & Johnson Interventional Systems (now Cordis) and was on the board of director of the first TAV company, Percutaneous Valve Technologies (PVT, now part of Edwards Lifesciences). Thubrikar has recruited him as its business advisor.
What Lies Ahead
Like many other developers of novel medical devices, Thubrikar Aortic Valve has decided to take its product to market through Europe initially, given European regulators’ comfort level with TAV and the FDA’s steep requirement for clinical trials. “We have spoken to the FDA and will continue to do so on a regular basis,” according to Dr. Thubrikar. “But they asked for a lot more preclinical testing than the European Notified Bodies to start a clinical trial.”
The company is now working to raise an additional $2 million to $10 million, and expects the granting of its patent for Optimum TAV in 2014. The finances will enable Thubrikar to not only conduct a first-in-human (FIH) feasibility study in up to 15 patients this year, but also to expand to a full European clinical trial of about 65 additional patients in 2015. If all goes well, a 2015 CE Mark for Optimum TAV isn’t out of the question.
However, trial success is vital, since today’s investors — and large companies in search of technology acquisitions — wait for significant clinical data to accumulate before backing a medical device. “We realize that until we actually implant the valve in a patient, other companies will think, ‘You don’t know what can go wrong,’” Dr. Thubrikar explained. “We had one big company say, ‘We will pay you four times as much once the product is in a patient.’ They want you to de-risk everything, to work out all the bugs yourself on your own dime.”
Yet Dr. Thubrikar thinks its only a matter of time until his life’s work finally arrives in the hands of interventional cardiologists, who he said have been “knocking at his door” since he first presented a paper on the technology in 2012. Since then, he has spoken at several of the largest interventional cardiology conferences, and word continues to spread about Optimum TAV. Like many other researchers-turned-entreprenuers, he steadfastly believes that his invention will eventually reach the market, where it can begin helping patients — like the one whose mother contacted him a decade ago.
“If hell freezes over, if we don’t get any money, I don’t care,” he said. “I don’t care how it happens. We are going to make a heart valve. That’s the only mission in my life.”
For more information on Thubrikar Aortic Valve and Optimum TAV, visit http://tavi.us/.
