Solving Early Stage Medtech Ventures' Funding Shortfall
By Eric Sugalski, Smithwise Medical Device Development

Gap funding panelists from the 2017 Pediatric Device Innovation Symposium included (from left) Nate Gross, MD, of Rock Health & Doximity; Kyp Sirinakis of Epidarex; Eric Sugalski (moderator) of Smithwise; Andrew ElBardissi, MD, of Deerfield; and Andrew Parker of Springhood Ventures
At the 2017 Pediatric Device Innovation Symposium established by the Sheik Zayed Institute for Pediatric Surgical Innovation at Children’s National Health System, a diverse group of investors came together to discuss gap funding for pediatric innovation. It is well-known within the industry that early stage medtech financing has been dwindling in the past decade. The critical phase of translational research, which exists between grant-funded academic research and institutional investment, is particularly lacking.
Fig. 1 highlights the funding gap that exists for the life sciences industry. While the data used to generate this graph included both biotech and medtech, the trends illustrated here seem to hold true for the medtech sector alone.
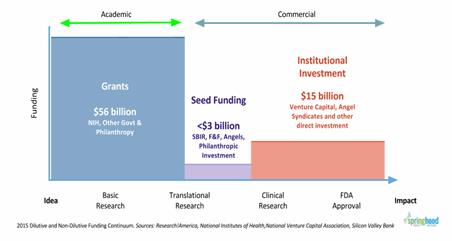
Fig. 1 — Funding Summary for Life Science Investments
The symposium’s gap funding panel candidly analyzed the current state of early stage medtech investment, but, more importantly, it concocted a plan of action and discussed a series of recommendations for key contributors to the early stage medtech ecosystem. Below is a recap of the panel’s discussion.
The Role of Clinical Innovators
Unmet clinical needs are most often identified by physicians and nurses; early stage concepts and technologies are often pioneered by these same medical professionals. However, these clinical innovators, due to lack of time and resources, are unable or unwilling to invest in new medtech ventures. That said, clinical innovators are crucial to the flow of new medtech opportunities, and their contributions directly affect the quantity and quality of future medtech deals. Clinical innovators also can play a vital role in bridging the early stage funding gap if they:
- Understand the Long Game — Often, clinical innovators believe that quick wins can be attained through a patent, a prototype, and a license deal with a large medical device manufacturer. Rarely does an established medical device company license and commercialize nascent medical device concepts that are loaded with risk. Most device innovators need to de-risk their concepts in order to gain traction among larger medical device entities. This de-risking process can involve facilitating clinical studies to prove the efficacy and economic viability of devices, developing new technologies to achieve accuracy levels and acceptable cost structures, and navigating complex regulatory and reimbursement pathways.
This process is a long one, requiring many different skill sets. While clinical innovators rarely drive these processes, it is critical for these individuals to gain insight into the timelines, operations, and budgets required so expectations are aligned at the onset of new ventures.
- Team Up — Most successful medical device innovations arise from clinicians teaming up with technical and business talent. Deerfield Management’s proprietary analysis suggests that 15-25 percent of novel medical device technologies come from academic institutions, with the remaining coming from commercial incubators or serial entrepreneurs / management teams.
Onboarding the right team members can de-risk a venture from investors’ perspectives. The right team is able to produce a logical and capital-efficient process while providing a credible set of skills that will advance the new venture.
The Role Of Academic Centers
Many research universities and hospitals attempt to fill some of the funding gap through internal innovation efforts. In many ways, academic centers serve as some of the main facilitators and gatekeepers of medical device innovation. Academic centers can take the following steps to overcome the funding gap:
- Be the Matchmaker — Universities and hospitals have adopted hack-a-thons as ways to connect clinical, technical, and business talent. These connections are essential, but are typically short-lived. Matchmaking between clinicians, entrepreneurs, and technical talent should be a continuous process.
- Provide Support — While it is ideal to find seasoned serial entrepreneurs to take on and spin out new medical technologies from academic centers, these business leaders usually are in high demand and short supply. Academic centers often need to assist in making device opportunities “investor ready” before seasoned entrepreneurs are willing to take the reins. This assistance typically entails de-risking through building prototypes, thoroughly vetting IP, and mapping out regulatory pathways. Many academic centers have created innovation funds to facilitate such de-risking processes. When internal resources are limited, though, innovation funds can be used to contract industry experts who can efficiently perform these risk reduction processes.
- Be the Dealmaker – While short-term licensing revenues tend to be the metric by which university tech transfer offices are evaluated, the self-reinforcing entrepreneurial ecosystems — as well as brand and reputation gains through innovative initiatives and future-entrepreneurs-turned-donors — can be far more valuable to academic centers in the long run. Implementing unreasonable terms for licensing new technologies is one of the greatest barriers to growth in the medtech sphere. Recognizing the immense value that entrepreneurs and investors can create post-licensing, and structuring appropriate terms that reflect this future value creation, can directly increase the number of opportunities and the investments that follow.
The Role of Entrepreneurs
Entrepreneurs driving new device ventures have many roles to play in strengthening the U.S. medtech industry.
- Fundraise to Specific Milestones — Early stage entrepreneurs facing funding challenges will often accept any funding that might help start their development process. Unfortunately, many of these entrepreneurs will face a downstream funding gap if sufficient progress has not been made through the prior financing round. When entrepreneurs are raising funds, they should do so with particular milestones in-mind.
These milestones should be identified to reduce specific risk(s), which in turn might attract future investors to enter a round at pricing levels that will satisfy prior investors and board members. Entrepreneurs also need to be realistic about the milestones they are able to achieve with early funding levels. Andrew Elbaridissi of Deerfield Management confirms, “In the current funding environment, aligning cash uses with the most significant drivers of value is crucial. Failure to do so places future financings and company viability at risk.”
- “Don’t be Wimps” – As suggested by CDRH Director Dr. Jeff Shuren, many entrepreneurs are excessively gun-shy about approaching regulatory bodies early in the process. The FDA and CMS offices are actively streamlining processes and creating new programs, such as Parallel Review for Regulatory & Reimbursement, which are not being pursued by entrepreneurs.
These programs are intended to help medtech entrepreneurs de-risk regulatory and reimbursement issues early in the process, and it’s in the best interest of entrepreneurs and the medtech ecosystem to participate in these reviews such that they remain in effect and continue to be optimized and expanded.
- Do your Homework — Epidarex’s Kyp Sirinakis emphasizes the importance of researching your investors. “You want ‘smart money,’ not just money, so make sure that you have knowledgeable investors,” she says. Additionally, the relationship matters. Srinakis compares it to getting married: “You will likely be working with these people for a long time through ups and downs. There needs to be mutual respect and the ability to communicate on both sides.”
- Plan, Plan, Plan — As John Parker of Springhood Impact Adventures explains, “Getting an innovative solution to the bedside is a long and difficult journey. It needs to be mapped out from start to finish, with the understanding that there will be traffic jams, detours, and roadblocks along the way. Sometimes, even the destination itself will change.”
Sirinakis backs up Parker’s claim, stating, “device ventures always cost more and take longer [than expected], so plan accordingly.” Developing the roadmap and trying to understand the timing and cost of all potential steps are keys to risk management. Since entrepreneurs typically need to get to market on a shoestring budget and timeline, it is difficult to bounce back from mistakes. “It’s important to do your homework on investors,” adds Sirinakis.
- Get Help / Bring Talent to the Process — Getting help from mentors, advisors, board members, and industry experts can add value and challenge your assumptions. Parker asserts, “[knowledgeable] people want to help. Tap the wealth of the free or low-cost resources out there for device developers, such as the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) — it won’t cost you a lot.”
The Role of Investors
Early stage investors take on considerable risk due to clinical, regulatory, payer, and technical challenges. Consequently, these early stage investors are susceptible to “getting squashed,” or substantially diluted, during later-stage financing rounds. When considering these dynamics, it makes sense why few early stage investors for medical devices exist.
Some investors are taking a different approach to filling this gap. Impact investors, particularly foundations and other charitable organizations, primarily measure their investments by the number of lives saved or improvements in quality of life for specific patient populations. Consequently, these types of investors tend to lean towards ventures that are making a significant impact, rather than those that serve as an incremental solution.
“Just as no return-driven venture capitalist hopes to make only a little bit of money, no early-stage impact investor strives to only make a little bit of difference,” Parker states. “Mission-focused investors are still looking for an outsized reward for the risk they are taking - they are just looking for that reward to manifest as massively improved outcomes for patients and healthcare stakeholders.”
In addition to providing this vital gap funding, the early stage investment community can bolster the medtech industry in the following ways:
- Get your Hands Dirty — Some of the most successful medtech investors are actively involved in contributing to the ongoing operations of portfolio companies. They provide valuable business connections, optimize go-to-market strategies, and ensure that milestones are being cleared in the most efficient means possible. A large part of an early stage investor’s role is to ensure that their portfolio companies do not fall into funding gaps down the road. This means structuring early stage financing to achieve specific milestones that will allow subsequent financing rounds to be achieved.
- Utilize Investor Networks — Investors bring their valuable networks and industry contacts to the table. “This can not only help you as run into pitfalls,” Sirinakis suggests, “but it can help introduce potential acquires and partners… ultimately helping structure the right deal.”
The Role of Regulators
U.S. regulators play an important role in keeping the population safe and striving for economic efficiency in our healthcare system. Indirectly, they also are pivotal in bridging the medtech funding gap. The constraints, processes, and ambiguity imposed by our regulating bodies constrict investment interest. Below are some actions that regulating bodies can take to improve the funding gap:
- Provide Reimbursement Clarity & Guidance — The FDA has created extensive guidance documentation to address questions and confusion within the medtech community, often providing requirements and suggestions for particular devices. Although payment for new therapies and diagnostics represents the most significant risk for early stage medtech companies, CMS has yet to provide this same level of clarity and guidance.
If a similar level of guidance documentation published by the FDA were to be published by CMS, it would provide the clarity for venture capital firms to invest more heavily in the medtech sector. Elbardissi explains, “The failure to couple reimbursement and regulatory pathways has long been a headwind in the medtech sector. This is emphasized to an even greater extent in pediatric devices, where small patient populations and an inability to gain orphan-like pricing and reimbursement represent a monumental impediment to investor appetite — which is critical to drive innovation.”
- Incentivize Investment in Unmet Needs — Many patient populations with unmet needs could be addressed through medtech innovation. Pediatrics, for example, is one of the most underserved patient populations. The lack of research and resources available for pediatric populations often leaves physicians with few options besides using adult devices on this population. The FDA is well aware of these issues, and they have created pathways to streamline the regulatory process.
The Humanitarian Device Exemption (HDE) pathway is one initiative the FDA has implemented to stimulate device innovation for small patient populations. While some medtech companies have benefitted from this pathway, it has not created adequate incentives to stimulate investment in these small patient populations. Conversely, the Orphan Drug Act, a seven-year patent extension and a substantial tax credit for companies developing and commercializing drugs for rare diseases, has done a remarkable job in stimulating investment for ventures focused on underserved populations.
Fig. 2 illustrates the number of orphan drug designations since 1982. If a comparable program was enacted to create tax incentives and patent term extensions for medtech, a significant stream of investment would likely open. Removing the profit caps that currently exist for these small patient populations would also greatly incentivize activity in these sectors.
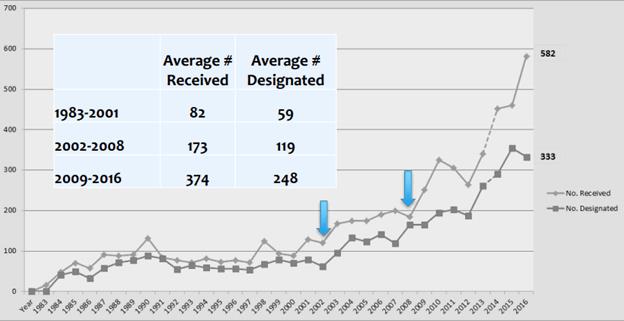
Fig. 2 — Orphan Drug Designations
The funding gap that currently exists for early stage medtech is a real threat to continued innovation within the medical device sector. While there is no simple fix to solving this complex funding challenge, there are specific actions that affiliated parties can take to improve the situation. Through discussions like those that occurred at the 2017 Pediatric Device Innovation Symposium, clinicians, entrepreneurs, academic institutions, investors, and regulators can connect, learn from one another, and plan for progress that will ultimately improve the U.S. medtech industry.
About The Author
Eric Sugalski is the founder and president of Smithwise, a contract medical device development firm with offices in Boston and Philadelphia. Sugalski has led the development of a novel pediatric life support system, cardiovascular implants, laparoscopic surgical devices, and an array of wearable diagnostics. In addition to his technical background, Eric provides companies with product development strategy that encompasses regulatory, reimbursement, and fundraising requirements. Eric obtained a B.S. in mechanical engineering from the University of Colorado Boulder and an MBA from the MIT Sloan School of Management.
