Look, Ma! How I Solved These Implantable Device Challenges
By Shawn Thibeault, Accumold
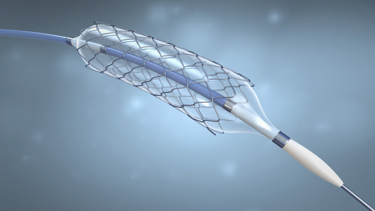
Designing and manufacturing implantable medical devices is a process highly regulated at every turn. Material choices, component quality, and a reliable supply chain also take on heightened importance when patient quality of life or physical well-being is at stake.
As a result of this high-pressure environment, medical implantable makers are understandably wary of change, unpredictability, or ill-defined pathways to successful commercialization. Accordingly, vendors supplying components to such device makers must exhibit an exemplary command of applicable regulations, as well as the technical acumen, production capacity, and sustainability necessary to thrive in a market where every risk is amplified.
Implantable Device Design Challenges
As noted above, medical device classification exposes products to a bevy of regulations that differ from country to country and some vendors lack the in-house familiarity to keep up with these ever-evolving rules. Vetting vendors to determine their grasp of these regulations is among the first steps a manufacturer embarks on when designing an implantable device.
Examined parallel to compliance prowess is a vendor’s material expertise and experience. For example, certain processes must be implemented if a part or a device is to be bioabsorbable (rather than a more traditional implantable) to ensure the material is not contaminated at any point during production, shipping, or mating to other components.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Med Device Online? Subscribe today.
