Package With A Purpose
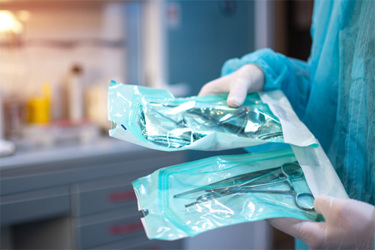
Medical device packaging shapes user behavior long before a device is ever handled. From storage decisions to unboxing and first use, packaging acts as the first user interface — communicating safety‑critical information, reinforcing correct mental models, and helping prevent use errors. When packaging is treated as an afterthought, manufacturers risk missed warnings, improper handling, and costly usability or regulatory setbacks.
This piece explores how a human‑centered approach transforms packaging into an active risk‑mitigation tool. It highlights practical design principles such as making critical information visible before opening, guiding intuitive opening and device removal, anticipating real‑world pharmacy and clinical environments, and aligning with regulatory expectations. Grounded in human factors and usability engineering standards, the discussion shows how packaging decisions directly influence safety, performance, and compliance.
Real‑world case studies illustrate how thoughtful packaging redesign can reduce dosing errors, prevent device damage, and counter incorrect user assumptions — often without changing the device itself. For teams developing or refining medical devices, this perspective reframes packaging as a strategic asset that supports safer, more effective use from the very first interaction. Read the full asset to explore actionable best practices and examples.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Med Device Online? Subscribe today.
